InnoGenic™ Non-resorbable Membrane
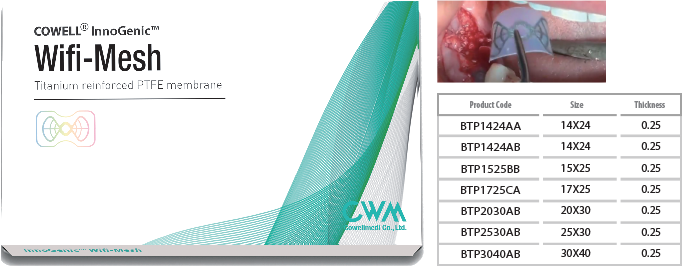
InnoGenic™ Wifi-Mesh and InnoGenic™ PTFE-Mesh
- The InnoGenic™ Wif i-Mesh, PTFE-Mesh and Ti-Mesh are non-resorbable barrier membrane to be applied over intraoral defects, especially, tooth extraction and bone augmented sites. The InnoGenic™ Wif i-Mesh and PTFE-Mesh are made of proprietary 100% PTFE, the polytetrafluoroethylene (teflon) sheet which is a biologically inactive and tissue compatible material and the InnoGenic™ Wif i-Mesh is reinforced with titanium frames (Titanium Gr II, ASTM F 67) embedded between two layers of PTFE sheets.
InnoGenic™ Wifi-Mesh
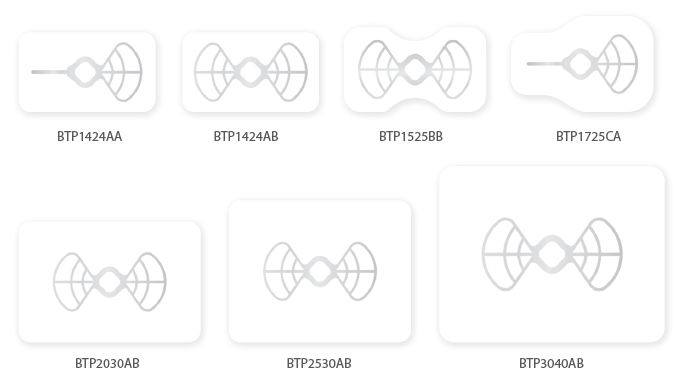
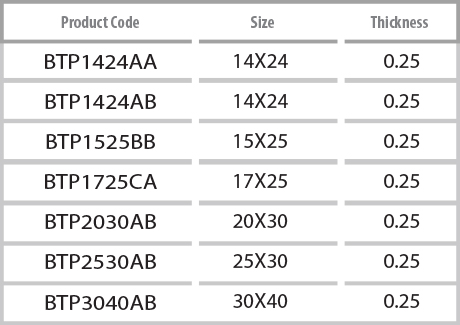
- Packing unit : 1ea

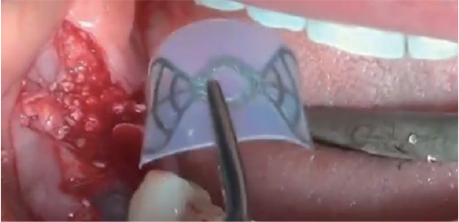
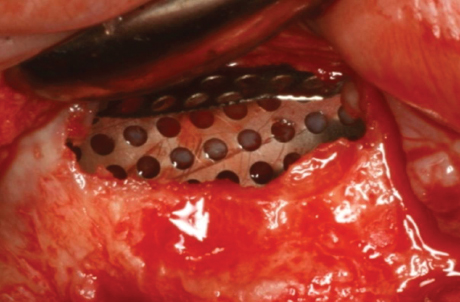
Clinical Case using the Wifi-Mesh
-
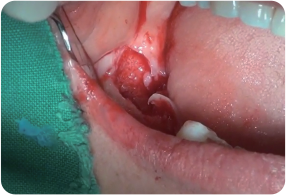
Periodontitis with local osteomyelitis of #45 & 47
-

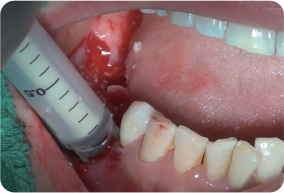
Bone graft using INNO-OSS™ Allo
-

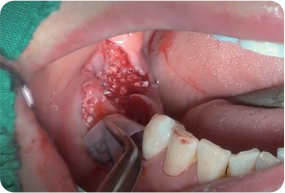
Shielding soft tissue penetration using Wifi-Mesh
-
3 months later
-

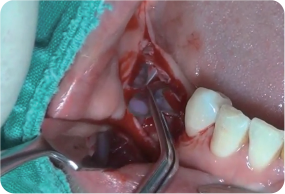
Removal of Wifi-Mesh
-

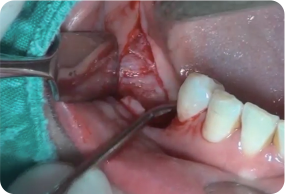
Dense periosteum layer has been formed
InnoGenic™ PTFE-Mesh
- Packing unit : 5ea
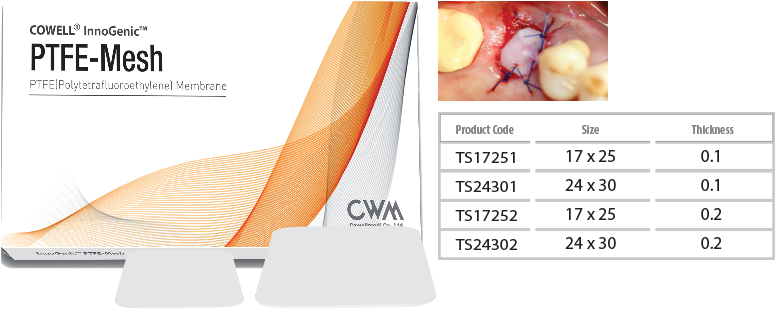

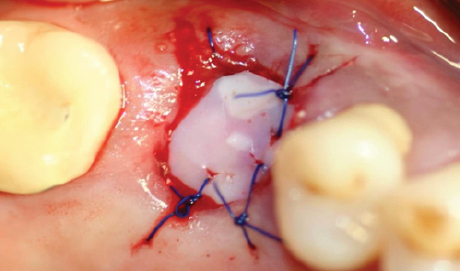

Features
- Non-resorbable : Made of 100% non-resorbable material for users to modulate healing period.
- Non-porous (0.0 μm) + Open Membrane Sheet Technique : Prevention of infection or other possible defects caused from passage or integration of bacteria through porosity of plaster and it even allows to apply Open Membrane Sheet Technique.
- Prevention of Displacement : Not only being sutured along with gingival but also using BOSS® Abutment and Screw inserted and fixed into the hall of titanium frame allows displacement of the products.
- Close to Transparency : Observation of the healing of the underlying tissue through almost transparent PTFE surface allows more predictable result and helps determine removal time easier.
- Easy to be Customized : Easy to modify the shape according to shape and dimension of the defect.
- Easy to be Removed : Put a hook in the hole of the titanium frame of the InnoGenic™ Wifi-Mesh and in any center part of the InnoGenic™ PTFE-Mesh and remove.
Comparison to other similar products sold in market
| Classification | Product A | Product B | InnoGenic™ Wifi-Mesh & PTFE-Mesh |
| SEM Photograph |


|


|


|
| Ultrastructure | Fiber | Filter | Sheet |
| Bacterial infection at exposure | Bacterial toxin penetration between filters at 50 μm intervals | Bacterial toxin penetration between filters at 2 μm intervals | No Bacterial toxin penetration thanks to non porous structure |
| Action on Exposure | Instant Removal | Removal on week 3 to 4 | Safe for more than 6 weeks |
| Shielding Function against Fiber Cell | High | High | Extremely High |
| Shape-keeping Capability against External Force | Large Deformation | Shrinkable Deformation | No Deformation |
InnoGenic™ Ti-Mesh
- The InnoGenic™ Ti-Mesh is made of stamping titanium sheet, also Titanium Gr II, ASTMF 67, which is 100% commercially pure titanium. The InnoGenic™ Ti-Mesh is non-resorbable surgical mesh to be applied over intraoral defects, especially, tooth extraction and bone augmented sites.
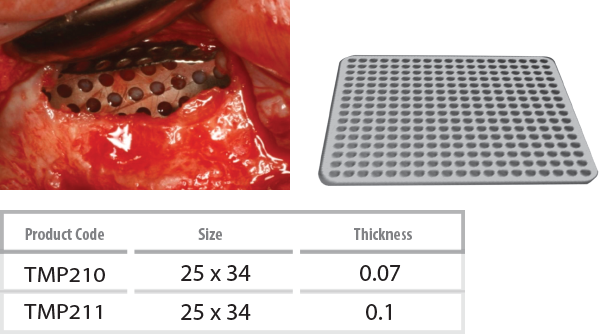

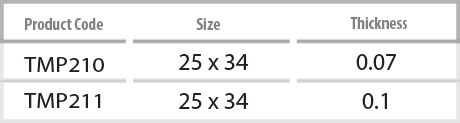
Features
- Easy to be Customized : Easy to modify the shape according to shape and dimension of the defect
- Prevention of Displacement : Prevents displacement of the InnoGenic™ Ti-Mesh using the BOSS® Abutment and Screw inserted and fixed into the 1mm hole of the Ti-Mesh Frame.
- No Memory : The problem of Majority of Titanium Meshes in the market is resilience of the products after certain time.
Due to this problem, patients go through serious pain. The InnoGenic™ Ti-Mesh is, however, made after many times of stamping process, the InnoGenic™ Ti-Mesh does not come back to the original shape after shape is formed.



